Lactic Acid - USP
Propanoic acid, 2-hydroxy-; Lactic acid CAS RN®: 50-21-5.
Lactic Acid is a chemically defined substance with specific purity requirements. This section details the characteristics, identification, testing, analysis, and labeling criteria for Lactic Acid.
Chemical Formula: C₃H₆O₃
Molecular Weight: 90.1
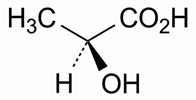
Definition
Lactic acid is a mixture of lactic acid (C₃H₆O₃) and lactic acid lactate (C₆H₁₀O₅), equivalent to a total of NLT 88.0% and NMT 92.0%, by weight, of lactic acid (C₃H₆O₃). It is obtained by the lactic fermentation of sugars or is prepared synthetically. Lactic acid obtained by fermentation of sugars is levorotatory, whereas that prepared synthetically is racemic.
Note: Lactic acid prepared by fermentation becomes dextrorotatory on dilution, which hydrolyzes L-(−)-lactic acid lactate to L-(+)-lactic acid.
Assay
Procedure
Sample: 2.5 mL, accurately weighed
Titrimetric system
- (See titrimetry (541).
- Mode: Residual titration
- Titrant: 1 N sodium hydroxide VS
- Back-titrant: 1 N sulfuric acid VS
- Endpoint detection: Visual
Analysis: Transfer the sample to a tared 250-mL flask, add 50.0 mL of titrant, and boil the mixture for 20 min. Add phenolphthalein TS, and titrate the excess alkali in the hot solution with back-titrant. Perform a blank determination. Each mL of titrant is equivalent to 90.08 mg of lactic acid (C₃H₆O₃).
Acceptance criteria: 88.0%–92.0% (w/w)
